|
Additional Notes: In 1755 Joseph Black in Edinburgh Scotland recognized magnesium as an element, however, it wasn't issolated until 1808 by Sir Humphrey Davy.Also used in fireplace bricks, flashbulbs, pigments and filters. Uses of Magnesium: Used in alloys to make airplanes, missiles, racing bikes and other things that need light metals.Primary mining areas are Austria, China, Poland, Russia, USA, India, Greece and Canada. World production is around 350,000 tons per year. Sources of Magnesium: Usually obtained by electrolysis of melted magnesium chloride (MgCl 2) found in sea water.Name Origin: Greek: From Magnesia a district of Thessaly.Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Vapor Pressure = C Regulatory / Health.Enthalpy of Vaporization: 128.7 kJ/mole.Enthalpy of Atomization: 148.5 kJ/mole 25☌.Reacts with hot water and burns in air when ignited. Conductivity Electrical: 0.226 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 26.1E -6.Valence Electron Potential (-eV): 40 Physical Properties of Magnesium. 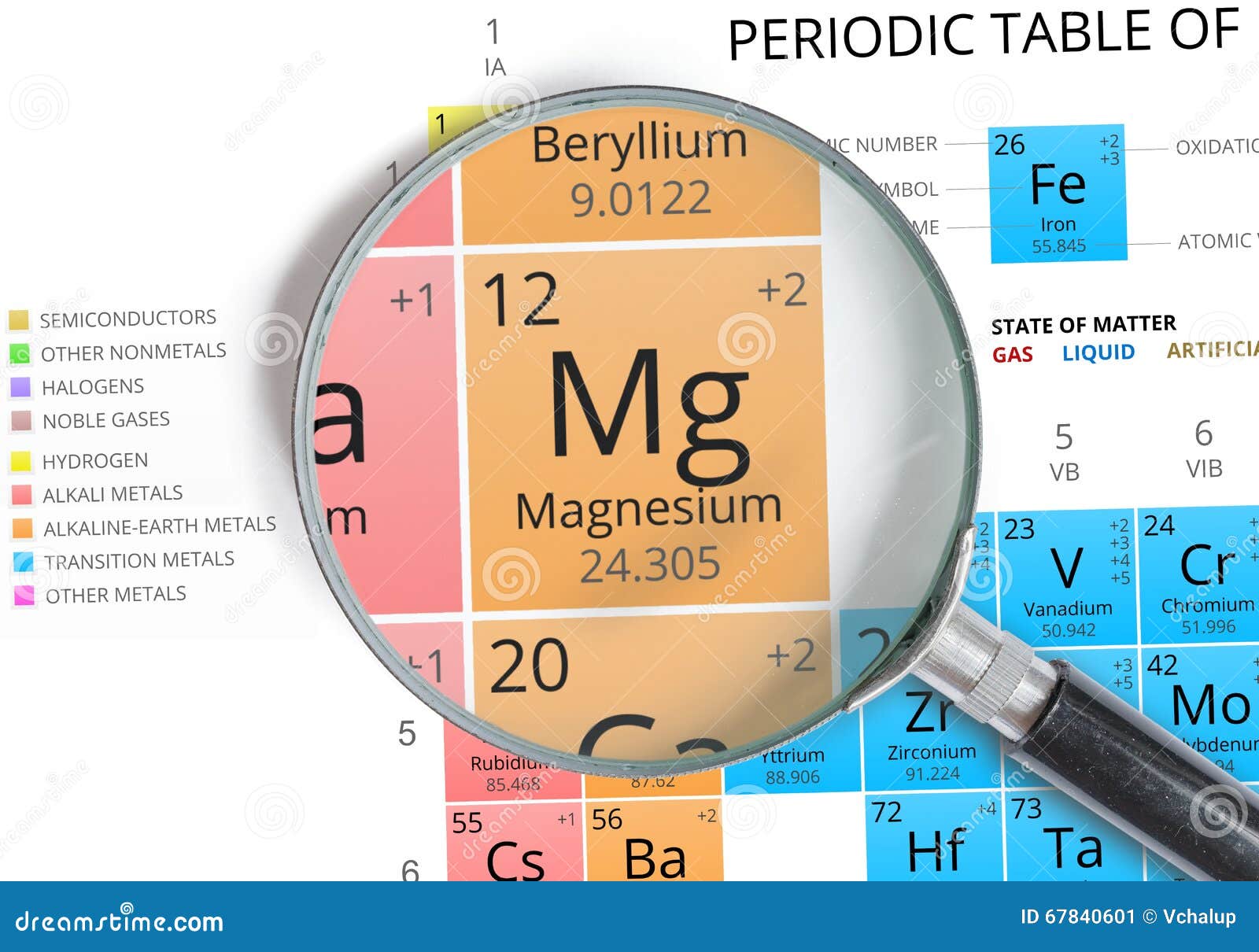
Electronegativity: 1.31 (Pauling) 1.23 (Allrod Rochow).Electrochemical Equivalent: 0.45341g/amp-hr.Valence Electrons: 3s 2 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 12. 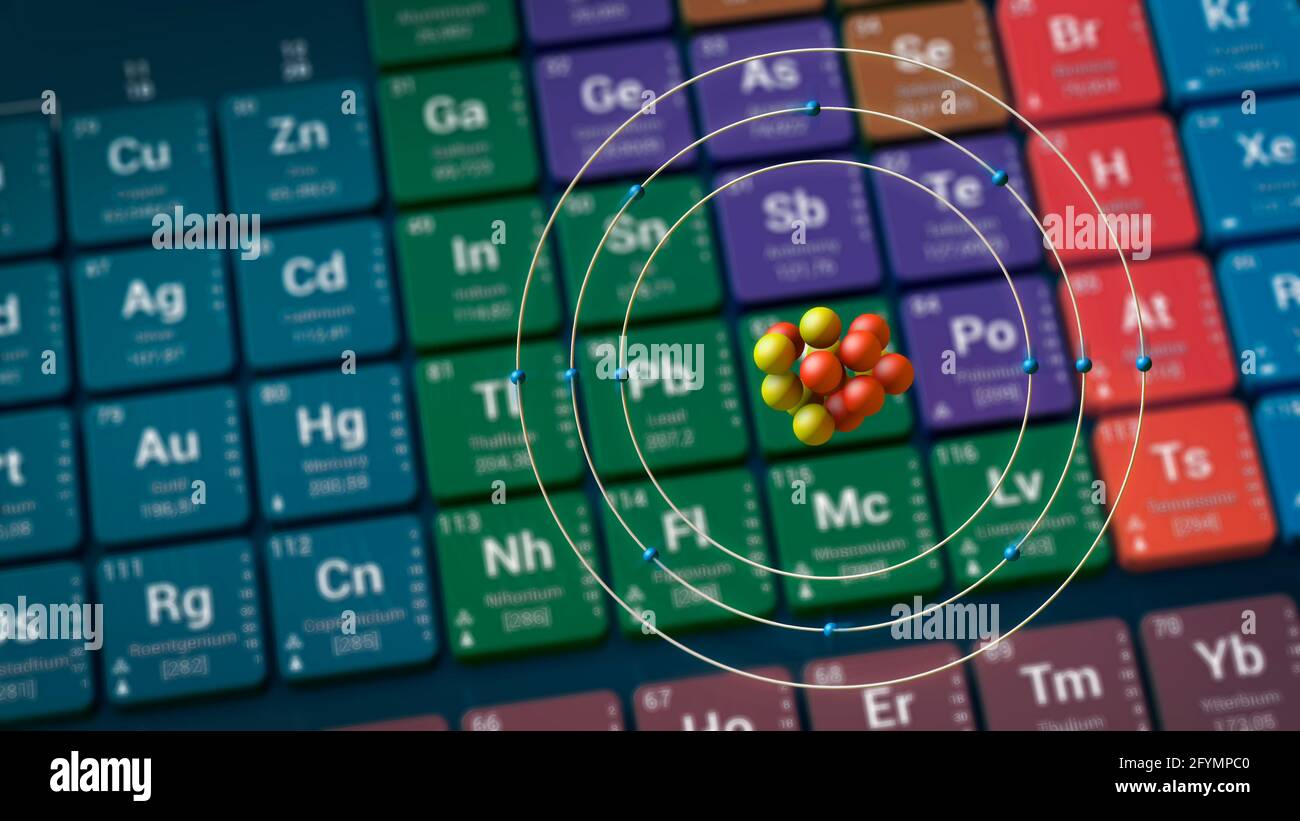
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
